PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
APPLICATIONS
APPEARANCE
ASSAY
pH
HEAVY METALS
20ppm max
5kgs - 25kgs
|
|
|
| CEFUROXIME SODIUM | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 56238-63-2 |
|
| EINECS NO. | 260-073-1 | |
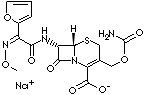
| FORMULA | C16H15N4O8SNa | |
| MOL WT. | 446.38 | |
|
H.S. CODE |
2941.90 | |
|
TOXICITY |
|
|
| SYNONYMS | Sodium cefuroxime; Zinacef; | |
| (6R,7R)-3-carbamoyloxymethyl-7-[2-(2-furyl)-2-[(Z)-methoxyimino]acetamido]-8-oxo-5-thia-1- azabicyclo[4.2.0]oct-2-ene-2-carboxylate, monosodium; Sodium [6R-[6alpha,7beta (Z)]]-3-[[(aminocarbonyl)oxy]methyl]-7-[2-furyl(methoxy imino)acetamido]-8-oxo-5-thia-1 -azabicyclo[4.2.0]oct-2-ene-2-carboxylate; Natrium-[6R-[6alpha, 7beta (Z)]]-3-[[(aminocarbonyl) oxy] methyl]-7-[2-furyl(methoxy imino)acetamido]- 8-oxo-5-thia-1-azabicyclo[4.2.0]oct- 2-en-2-carboxylat; [6R-[6alpha,7á (Z)]]-3-[[(aminocarbonil)oxi] metil]-7-[2-furil(metoxi imino)acetamido]- 8-oxo-5-tia-1- azabiciclo[4.2.0] oct-2-eno-2-carboxilato de sodio; [6R-[6alpha,7beta (Z)]]-3-[[(aminocarbonyl) oxy]méthyl]-7-[2-furyl(méthoxy imino)acétamido]-8- oxo-5-thia-1-azabicyclo [4.2.0]oct-2-ène-2- carboxylate de sodium | ||
|
CLASSIFICATION |
||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | White to slightly yellowish powder | |
| MELTING POINT | ||
| BOILING POINT | ||
| SPECIFIC GRAVITY | ||
| SOLUBILITY IN WATER | ||
| pH | ||
| VAPOR DENSITY |
| |
|
AUTOIGNITION |
| |
|
NFPA RATINGS |
||
|
REFRACTIVE INDEX |
||
| FLASH POINT |
| |
| STABILITY | Stable under ordinary conditions. | |
|
APPLICATIONS |
||
| Cephalosporin: any of a group of broad-spectrum derived from species of fungi of the genus Cephalosporium and are related to the penicillins in both structure and mode of action but relatively penicillinase-resistant antibiotics. These antibiotics have low toxicity for the host, considering their broad antibacterial spectrum. They have the active nucleus of beta-lactam ring which results in a variety of antibacterial and pharmacologic characteristics when modified mainly by substitution at 3 and 7 positions. Their antibacterial activities result from the inhibition of mucopeptide synthesis in the cell wall. They are widely used to treat gonorrhea, meningitis, pneumococcal, staphylococcal and streptococcal infections. The cephalosporin class of antibiotics is usually divided into generations by their antimicrobial properties. Three generations of cephalosporins are recognized and the fourth has been grouped. Each newer generation of cephalosporins has broader range of activity against gram-negative organisms but a narrower range of activity against gram-positive organisms than the preceding generation. The newer agents have much longer half-lives resulting in the decrease of dosing frequency. Accordingly, the third-generation cephalosporins can penetrate into tissues well, and thus antibiotic levels are good in various body fluids. Second-generation cephalosporins have broader spectrums of activity against gram negative coverage but less active against gram-positive organisms than first-generation agents. This group includes cefamandole (CAS RN 34444-01-4), cefaclor (53994-73-3), cefuroxime (55268-75-2), cefonicid (61270-58-4), ceforanide (60925-61-3). Cefoxitin (35607-66-0) and cefotetan (69712-56-7) which are effectively active against bacteroides fragilis are also included in this group, although they are cephamycins actually. | ||
| SALES SPECIFICATION | ||
|
APPEARANCE |
White to slightly yellowish powder | |
|
ASSAY |
855 - 1,002µg/mg | |
| MOISTURE | 3.5% max | |
|
pH |
6.0 - 8.5 | |
|
HEAVY METALS |
20ppm max |
|
| TRANSPORTATION | ||
| PACKING | 5kgs - 25kgs | |
| HAZARD CLASS | ||
| UN NO. | ||
| OTHER INFORMATION | ||
|
|
|